
What's the difference between an acid gas and organic vapor?
I know there are a variety of air purifying respirator cartridges available, but the two most commonly used (non-particulate) ones seem to be the acid gas and organic vapor cartridges.
I know the spec sheets will normally list out the specific compounds each protect against, but can someone explain the general difference between an acid gas and organic vapor? I hate relying on the spec sheets every time - would be nice if I had a fundamental understanding XD I'm also curious how the cartridges actually remove the compounds (if anyone knows)
Comments (10)

This is why I generally spend a few extra bucks and get the multichemical/P100 cartridge, haha. They do also sell OV/Acid gas combo cartridges. Most of the cartridges have an absorbent (e.g., activated carbon) inside to filter out the chemicals, but there are other methods of filtration, as well.
Generally speaking, organic vapors refers to petroleum-based chemicals that have a high vapor pressure (evaporates readily and quickly). Acid gases, on the other hand, deals specifically with gases produced from corrosive (acidic) chemicals and creates an acidic solution when mixed with water.

That’s actually a tough one, even for folks like me who specify respiratory protection all the time.
Organic vapor is easy, as it would be any volatile organic compounds, which are carbon based chemicals.
Acid gas is tough, because even if you memorize your inorganic acids, the chemical name in a gas might be different than the one you’ve memorized in a solution.

Organic vapors are vapors that produced by volatile organic compounds (VOC). These compounds are hydrocarbons and their derivatives. Examples include things like toluene, xylene, acetone and other solvents (there are thousands of hydrocarbon derivatives). Compounds with high vapor pressure create more vapors.
Acid gases are gases that when dissolved in water create acidic solutions. Most common acid gases are hydrogen fluoride, hydrogen chloride, and hydrogen bromide. There are other gases the create acidic solutions but I have not heard them called acid gases. These include oxides of sulfur (acid rain component), oxides of nitrogen (fuming nitric acids), and carbon dioxide (carbolic acid, it's why soda is acidic) are the most common.
When an acid gases is bubble through water, it creates an acidic solution. For example, when hydrogen chloride gas is bubble through water, some of the gas dissolves in the water. This solution is called hydrochloric acid (also muriatic acid). The gas often will come out of solution and can be inhaled. When it enters the lungs, it will mix with the moisture in the lungs to create an acidic solution again.
Ammonia is also a gas that is often dissolved in water, however, it creates an alkaline solution rather than an acid.

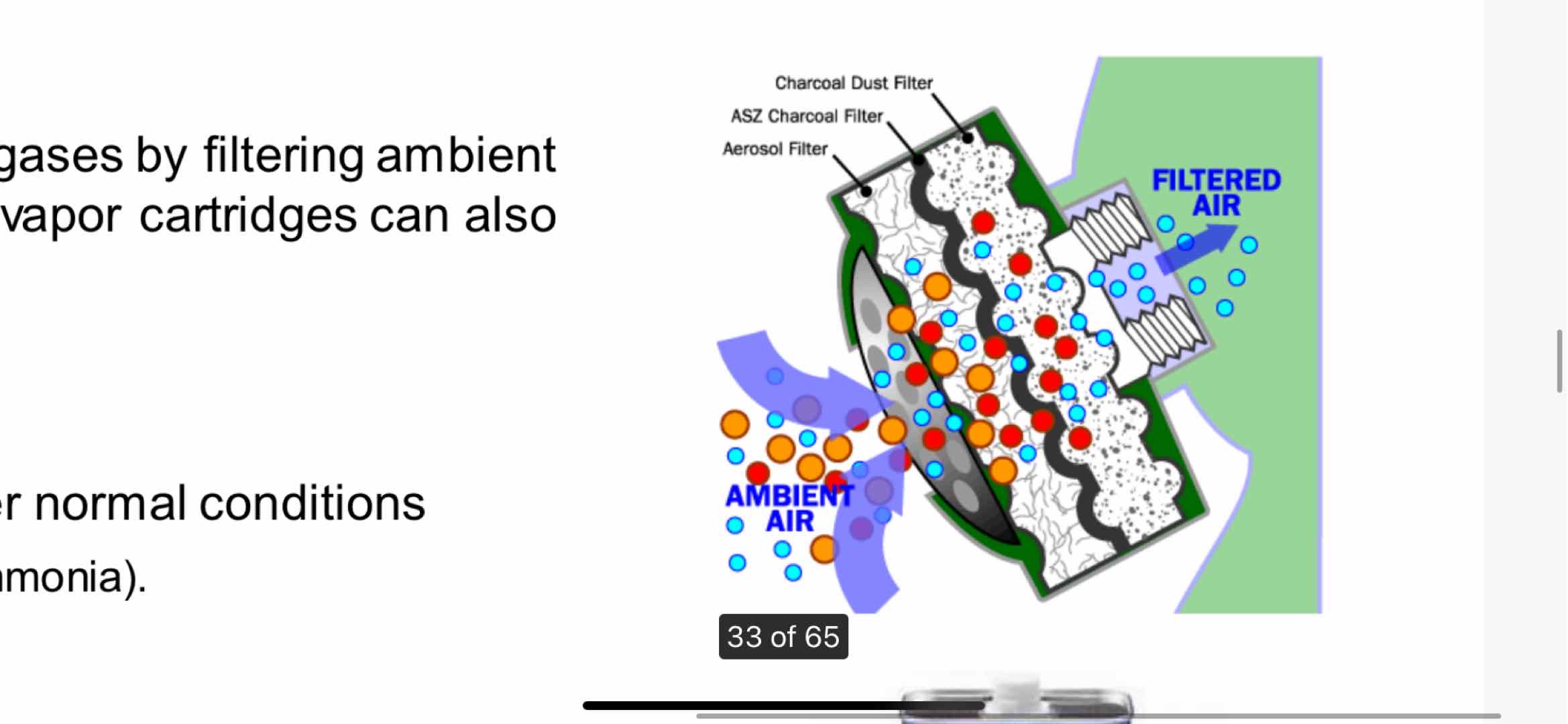
Most manufacturers use activated carbon. Here is a rudimentary diagram of how the filter works.

Here is the north cartridge reference chart. Anyone can send me your email address and I can share the full document. Of course this is just for north filters. We did recently add a nitrogen dioxide filter as well
